中国科学院微生物研究所、中国微生物学会主办
文章信息
- 袁建华, 赵天涛, 彭绪亚
- Yuan Jianhua, Zhao Tiantao, Peng Xuya
- 极端条件下异养硝化-好氧反硝化菌脱氮的研究进展
- Advances in heterotrophic nitrification-aerobic denitrifying bacteria for nitrogen removal under extreme conditions
- 生物工程学报, 2019, 35(6): 942-955
- Chinese Journal of Biotechnology, 2019, 35(6): 942-955
- 10.13345/j.cjb.180427
-
文章历史
- Received: October 17, 2018
- Accepted: March 5, 2019
2. 重庆理工大学 化学化工学院,重庆 400054
2. College of Chemistry and Chemical Engineering, Chongqing University of Technology, Chongqing 400054, China
氨氮超标排放会导致水体黑臭及富营养化,不仅对鱼类及水生生物产生毒害作用,而且严重危害生态平衡及人体健康[1]。传统生物脱氮技术具有条件温和、处理成本低、二次污染小等优势,但随着污水排放标准提高,传统生物脱氮技术已无法满足可持续发展需求[2-3]。尤其在应对极端条件废水处理时,该类技术暴露出生物活性低、反应速度慢、处理效果差等技术瓶颈[4]。常见极端条件主要包括高盐、低温、高氨氮等。高盐废水通常是指盐度超过1%的废水,过高的盐度会引起细胞酶活降低,丧失脱氮功能[5];低温条件在我国比较普遍,很多废水处理设施处于高海拔、高纬度的低温地区,当温度每降低10 ℃时,会导致脱氮微生物的酶促反应速率降低1–2倍[6];高氨氮废水是指氨氮浓度超过300 mg/L的废水,污泥脱水液、厌氧消化液、垃圾浓缩液等都属于此类废水。高浓度的游离氨对硝化菌具有强抑制和高毒害作用,现有工艺多采用投加酸碱等化学试剂进行控制,易造成废水处理费用高昂、二次污染等问题[7]。应对这些极端条件,开发经济高效的新型生物脱氮技术一直是该领域的研究热点。
1983年,Robertson等从脱硫脱硝废水中首次分离得到了异养硝化-好氧反硝化(HN-AD)菌株泛养副球菌Paracoccus pantotrophus,它主要包括氨单加氧酶(AMO)、羟胺氧化酶(HAO)、硝酸还原酶(NAR)等功能酶[8]。HN-AD菌属在好氧条件下可快速将氨氮、硝态氮(NO3–-N)、亚硝态氮(NO2–-N)转化为含氮气体,整个代谢过程几乎没有NO3–-N和NO2–-N积累[9]。研究者已筛选得到了约20个属近100多种具有HN-AD代谢特性的菌株,这些菌株来源于土壤、污泥、水体以及火山口、盐碱地等环境中[10]。随着对HN-AD的深入研究,科研人员惊喜地发现HN-AD菌具有环境适应能力强、污染物耐受浓度高等优点。如Lei等得到的台湾佐氏菌Zobellella taiwanensis DN-7耐受最高氨氮浓度可达2 000 mg/L[11];Duan等筛选的嗜盐弧菌Vibrio diabolicus SF16在盐度为5%时,对120 mg/L的氨氮去除率达到了92.1%,总氮(TN)去除率达到了73.9%[12];Yao等分离的不动杆菌Acinetobacter sp. HA2在温度低至10 ℃时,可将80 mg/L的氨氮和TN都100%去除[13]。除此之外,HN-AD菌属还具有世代时间短、脱氮速率快、高活性持久等优势,这使其在极端条件废水处理领域表现出了巨大的应用潜力[14]。这些研究从菌属种类、脱氮能力及机理方面对HN-AD菌的特性进行了初步探索,然而关于HN-AD菌在极端条件下脱氮的关键基因和代谢机制的解析还不全面,不同极端条件下的菌种投加方式、碳源选取类型、工艺优化参数等尚未完全阐明。系统归纳HN-AD菌属在极端条件的脱氮进展将有助于丰富HN-AD脱氮理论和推进HN-AD脱氮工艺的研发,而目前此类综述鲜有报道。
据此,文中对极端条件下具有脱氮性能的HN-AD菌属开展广泛调研,系统总结国内外关于HN-AD菌属在高盐、低温、高氨氮等极端条件脱氮的研究成果,对比分析不同HN-AD菌属的耐受程度及脱氮特性;并结合课题组在HN-AD菌属筛选及脱氮研究方面的进展,考察该类菌属在极端条件下的应用效果;最后对HN-AD脱氮技术工程的应用前景及研究方向进行了展望。以期为HN-AD菌属进行极端条件下废水氨氮的生物脱除提供理论依据和技术支撑。
1 HN-AD菌属及代谢特性自1983年Robertson等首次分离得到了异养硝化-好氧反硝化(HN-AD)菌株Paracoccus pantotrophus[8] (曾用名Thiosphaera pantotropha)以来,目前,约20多个属的HN-AD菌株被分离出来,主要有产碱菌属Alcaligenes、假单胞菌属Pseudomonas、芽孢杆菌属Bacillus、不动杆菌属Acinetobacter等。表 1对近十年不同种属菌株的来源、碳源、碳氮比(C/N)等进行了总结。归纳菌株的来源,发现HN-AD菌除了存在于土壤、污泥、废水中,还存在于火山岩浆、盐碱湿地、低温冻土等极端环境中[10],由此表明HN-AD菌不仅分布广泛,而且对极端环境具有耐受能力;分析所需的碳源,可知HN-AD菌属能快速利用葡萄糖和蔗糖等易吸收的糖类碳源,也能利用柠檬酸钠、琥珀酸钠、丁二酸钠等非糖类碳源。总体上,不同种属的HN-AD菌对碳源呈现出一定的选择性[15];对于C/N,HN-AD菌属的适应范围较宽,通常在6–20之间,多数菌株需求的C/N在10左右,少数菌株能耐受C/N≤5的贫营养条件[16]。
| Genus | Strain | Source | Carbon source | Carbon to nitrogen ratio | Ammonia removal (%) | References |
| Halomonas | X3 | Aquaculture wastewater | Sodium acetate | 10 | 80.0 | [20] |
| Arthrobacter | N6 | soil | Sodium citrate | 5 | 80.0 | [21] |
| Rhodococcus | CPZ24 | Pig farm | Sodium succinate | Not mentioned | 100.0 | [22] |
| Aeromonas | HN-02 | Activated sludge | Sodium succinate | Not mentioned | 80.8 | [23] |
| Alcaligenes | NR | Membrane bioreactor | Glucose | Not mentioned | 100.0 | [24] |
| Serratia marcescens | N-2 | Culture pond | Sucrose | Not mentioned | 90.8 | [25] |
| Pseudomonas | YZN-001 | Pig manure wastewater | Sodium succinate | 10 | 95.0 | [26] |
| YG-24 | Lake sediment | Sodium citrate | 8 | 88.1 | [27] | |
| AND-42 | Sponge | Not mentioned | 12 | 75.8 | [28] | |
| Acinetobacter | Y16 | Not mentioned | Sodium acetate | 2 | 66.0 | [29] |
| Sxf | Reservoir Sediment | Sodium succinate | 1 | 65.6 | [30] | |
| YB | Activated sludge | Sodium succinate | 15 | 99.3 | [31] | |
| YF14 | Pond | Sodium succinate | 8 | 92.0 | [32] | |
| A14 | Not mentioned | Sodium acetate | 12 | 95.3 | [33] | |
| Diaphorobacter | PD-7 | Laboratory | Not mentioned | 8 | 100.0 | [34] |
| WS-2 | Landfill | Sodium succinate | Not mentioned | 94.7 | [35] | |
| Golden bacillus | R31 | Slaughter wastewater | Glucose/sodium succinate | 10 | 95.9 | [36] |
| Coctobacillus | S1 | Coking wastewater | Sodium pyruvate | 14 | 99.7 | [37] |
研究者不但开展了大量HN-AD菌种的分离工作,而且对HN-AD的关键酶和代谢特性也进行了深入研究。1998年Richardson等[17]发现HN-AD脱氮过程的关键酶主要包括氨单加氧酶(AMO)、羟胺氧化酶(HAO)、周质硝酸盐还原酶(NAP)、亚硝酸还原酶(NIR)、一氧化氮还原酶(NOR)和一氧化二氮还原酶(NOS),接着又提出了目前国内外公认的脱氮代谢途径,如图 1所示,首先氨在AMO的作用下氧化生成羟胺,然后羟胺在HAO的作用下生成亚硝酸盐,最后亚硝酸盐或硝酸盐在NIR、NOR、NOS的还原作用下分别生成NO、N2O和N2,整个脱氮过程基本没有NO3–-N和NO2–-N的积累。
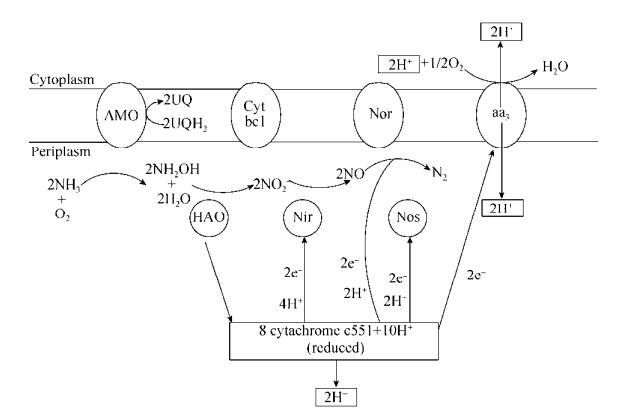
随后,许多学者对HN-AD提出了不同的代谢模型,其中以Wehrfritz等[18]提出的偶联模型最具代表性。如图 2所示,该模型以亚硝酸盐作为硝化反硝化偶联的分节点,异养硝化时羟胺被氧化为NO2–,所产生的电子直接传递给反硝化酶系的电子供体细胞色素c550,通过细胞色素c550先将电子传递给NIR、NOR、NOS,最后将电子传递给细胞色素aa3氧化酶将氧还原为水。异养硝化是指微生物利用有机物作为碳源同时将氨氮转化为羟胺、亚硝态氮、硝态氮等;好氧反硝化是指微生物在有氧气和碳源存在的条件下,利用O2和NO2–-N等作为电子受体进行的呼吸作用。HN-AD菌属特殊的代谢通路实现了硝化和反硝化在异养和好氧的条件下同时发生,打破了传统硝化反硝化必须分隔以及反硝化必须在缺氧条件下进行的限制,是对传统硝化反硝化理论的丰富和突破[19]。
总体上,HN-AD菌表现出了分布范围广、适应能力强、代谢通路特殊等特点。深入调研发现HN-AD菌属还具有世代时间短、脱氮速率快、高活性持久等独特优势,这使其在应对极端条件时具备了较高的耐受性,文中从高盐、低温、高氨氮等方面对耐受极端条件的HN-AD菌属进行了总结。
2 极端环境HN-AD菌属脱氮研究现状 2.1 高盐环境下HN-AD脱氮高盐废水一般是指总含盐量大于1%的废水,含有较高浓度的Cl–、SO42–、Na+、Ca2+等无机离子[38]。高盐废水来源广泛,主要包括印染、腌制、造纸等化工生产以及海水直接利用过程产生的废水[39]。我国高盐废水总量巨大,约占废水总量的5%,并每年以2%的速率增长[5]。生物法开始逐渐应用于高盐含氮废水处理,然而普通微生物处理高盐废水时由于盐度过高一直存在处理系统菌群数量减少、有机物和氨氮去除率降低、出水悬浮物浓度大等诸多问题[40-41],所以筛选耐盐、嗜盐的脱氮功能微生物逐渐成为高盐含氮废水处理领域研究的热点。
研究者在分离耐盐、嗜盐脱氮功能微生物的过程中,发现许多HN-AD菌同时表现出了耐盐和脱氮的性能。如Duan等[12]从海洋沉积物中筛选出一株嗜盐弧菌Vibrio diabolicus SF16在盐度为5%、氨氮浓度为120 mg/L的条件下,培养48 h后氨氮去除率达到了92.1%,TN去除率达到了73.9%;Chen等[23]研究的嗜水气单胞菌Aeromonas sp. HN-02在盐度为2%的条件下仍表现出较强的氨氮和TN去除能力,氨氮平均去除速率达30 mg/(L·h),24 h内TN去除率达到了80.8%,且1%的盐度对氨氮的去除基本没有影响;曲洋等[42]分离的假单胞菌Pseudomonas qy18和盐单胞菌Halomonas gs2的适宜生长盐度范围分别为0–4%和2%–10%,在48 h内菌株qy18和菌株gs2的氨氮去除率分别为98.5%和96.1%,TN去除率分别达到了83.6%和81.5%,最大氨氮去除速率分别为5.8 mg/(L·h)和5.5 mg/(L·h),两株菌在高盐度条件下可同时利用有机碳源和氨氮为底物进行硝化反应和同步脱氮。
根据不同菌属的耐盐能力差异,目前将耐受盐度小于3%的细菌定义为耐盐菌,在3%–15%之间的细菌称为中度嗜盐菌,大于15%的细菌称为极端嗜盐菌[43],表 2根据HN-AD菌属的耐盐能力对其进行了分类。由表 2可知,耐盐HN-AD菌属主要来源于海洋、盐场底泥、腌制废水等含盐环境,其耐盐和脱氮能力受分离场地影响较大,如从排污淤泥中筛选的施氏假单胞菌Pseudomonas stutzeri A-13耐受的盐浓度达到了10%,且对初始浓度分别为620 mg/L、570 mg/L、545 mg/L的NO3–-N、NO2–-N、氨氮去除率分别达到了99.7%、90.7%和67.7%,最高TN去除率达到了80.8%[44],而从生活废水中获得的甲养芽孢杆菌Bacillus methylotrophicus L7耐盐浓度仅为3.5%,最大氨氮去除率为83.4%,最大NO2–-N去除速率为5.81 mg/(L·d)[45]。HN-AD菌的耐盐浓度范围通常在5%–10%之间,属于中度嗜盐菌,某些特殊菌种能耐受超过10%的盐度,如盐田盐单胞菌Halomonas campisalis ha3[46],能够耐受20%的高盐度,其最大NO3–-N去除速率达到了87.5 mg/(L·h),与其他HN-AD菌株不同,它的最适pH为9.0,除了嗜盐,还表现出一定的嗜碱性。HN-AD菌属耐盐脱氮的特性,克服了高盐对传统生物处理过程的限制,同时解决了传统微生物耐盐不脱氮、脱氮不耐盐的矛盾[47],未来在海鲜加工、水产养殖、腌制等高盐废水脱氮处理方面具有较大的应用价值。
| Salt-resistant classification | Genus | Strain | Source | Highest salt tolerance (%) | Ammonia removal (%) | Total nitrogen removal (%) |
| Salt tolerance | Aeromonas sp. | HN-02 | Freshwater environment | 2 | Not mentioned | 80.8 |
| Mild halophile | Marinobacter sp. | F6 | Marine environment | 3 | Not mentioned[48] | 50.1 |
| Bacillus methylotrophicus | L7 | Wastewater | 4 | 39.5 | Not mentioned | |
| Bacillus cereus | X7 | Pickled wastewater | 4 | 97.6[49] | 52.1 | |
| Halomonas sp. | B01 | Salt field sediment | 6 | 99.20[50] | 96.0 | |
| Pseudomonas stutzeri | A-13 | Sewage sludge | 10 | Not mentioned | 86.1 | |
| Halophile | Halomonas campisalis | ha3 | Not mentioned | 20 | Not mentioned | Not mentioned |
常规脱氮菌株的最适温度范围为28–37 ℃,当温度每降低10℃,脱氮微生物的总量和酶活均受到严重影响,污染物降解效率降低1–2倍[6]。为了解决常规脱氮微生物无法耐受低温这一难题,分离在低温条件下具有高效脱氮功能的菌株越发受到研究与工程技术人员的关注[51-52]。
近年来,HN-AD菌属在低温条件下的脱氮潜能逐渐被挖掘。表 3根据耐低温程度对脱氮HN-AD菌株进行了总结。HN-AD菌属耐受低温的范围为2–15 ℃,其中耐受5–10℃的菌株较多,它们在低温条件下仍保持了较高的脱氮速率。如Yao等[13]筛选的不动杆菌Acinetobacter sp. HA2在10 ℃下,氨氮脱除速率可达到3 mg/(L·h),TN去除率达到了100%;恶臭假单胞菌Pseudomonas putida Y-12[53]在15 ℃时,4 d后能将初始氨氮浓度为208.1 mg/L脱除到2.4 mg/L,氨氮去除率达到98.8%,TN去除率平均达到75.5%以上;拟南芥假单胞菌Pseudomonas migulae AN-1[54]在10 ℃时,氨氮、NO3–-N、NO2–-N脱除效率分别可达到1.6 mg/(L·h)、1.5 mg/(L·h)和0.69 mg/(L·h);从松花江分离的微杆菌属Microbacterium sp. SFA13在温度为5 ℃、初始氨氮浓度为61.6 mg/L时,氨氮和NO3–-N的脱除速率最高分别达到了1.6 mg/(L·h)和0.24 mg/(L·h)[55]。课题组分离的HN-AD菌株TAC-1,在5 ℃的低温条件下、能将浓度为400 mg/L的氨氮快速脱除,去除速率达到了3.7 mg/(L·h),同时TN去除率达到了79.5%。归纳发现耐冷HN-AD菌属在5–10 ℃时,氨氮平均脱除速率为2–3 mg/(L·h),在10–15 ℃时通常为4–6 mg/(L·h),可见HN-AD菌属在低温条件下的脱氮速率在一定范围内随温度的升高而升高。总体而言,这些耐冷HN-AD菌株不仅在低温条件下进行正常的生长代谢,而且实现了氨氮和总氮的高效脱除,足以体现其具有应用于低温废水脱氮的潜能。
| Tolerable minimum temperature (℃) | Genus | Strain | Ammonia removal or degradation | Total nitrogen removal (%) |
| ≤5 | Acinetobacter sp. | Y16 | 66.0% | 62.1 |
| Aeromonas hydrophila | HN-02 | 2.6 mg/(L·h) | 80.8 | |
| 5–10 | Pseudomonas fluorescens[61] | wsw-1001 | 71.0% | 68.9 |
| Acinetobacter sp. | HA2 | 3.0 mg/(L·h) | 100.0 | |
| Pseudomonas migulae | AN-1 | 1.6 mg/(L·h) | Not mentioned | |
| Microbacterium sp. | SFA13 | 3.2 mg/(L·h) | 100.0 | |
| 10–15 | Arthrobacter arilaitensis[62] | Y-10 | 65.0% | 85.3 |
| Pseudomonas putida[63] | Y-9 | 2.9 mg/(L·h) | Not mentioned | |
| Pseudomonas putida | Y-12 | 9.8 mg/(L·h) | 32.1 | |
| Acinetobacter sp.[64] | DW4 | 65.0% | 100.0 |
为了适应低温环境,耐冷微生物通常会通过改变脂类组成来调节膜的流动性以适应温度变化,同时,它们还会生成冷休克蛋白和冷适应酶来应对低温环境[56];与传统分子机制不同的是,HN-AD菌在低温条件下还会产生较多的抗氧化酶以保护机体免受超氧阴离子、过氧化氢、OH自由基造成的损伤[57];大多数HN-AD菌对C/N的需求较高,多数菌株需求的C/N在10左右。在高浓度有机碳环境下,菌体自身好氧代谢会大量产热,热量一部分用于细胞合成和代谢,多余部分则以热的形式散发,其表现在宏观上使得外界温度升高[58-59];此外,当外界温度较低时,活性污泥和生物膜会调整自身的代谢产热系统,在相应减少细胞合成的同时,将大量ATP转化为热量,用于平衡和调节胞内和外界温度,以此相对提升自身的低温适应能力[60]。现有研究表明HN-AD菌属具有独特的耐冷机制,但基于HN-AD菌属细胞水平和基因水平的耐冷机制研究还较少,详细的代谢机理和关键基因还有待进一步确定。
2.3 高氨氮条件下HN-AD脱氮目前,国内外处理高氨氮废水的主要方法分为物化法、生物法、物化-生物联合法,对于污泥脱水液、厌氧消化液、焦化废液等可生化性差、氨氮浓度极高的废水,通常采用折点加氯、气提吹脱和离子交换等物化法进行处理[65];对于氨氮浓度在500–1 500 mg/L左右的废水,常采用物化-生物等多工艺联合进行处理,但物化法作为前端处理此类废水时普遍存在运行费用昂贵的缺点。传统生物脱氮技术易受游离氨影响,处理废水的氨氮浓度不宜超过300 mg/L,虽然游离氨的控制技术已比较成熟,但脱氮过程中大量碱度的消耗及曝气等电耗同样导致处理成本高昂,所以目前利用传统微生物进行高氨氮废水的处理仍然存在瓶颈[66-67]。因此,筛选对高氨氮具有耐受和脱除能力的功能微生物是未来处理此类废水的研究方向[68]。
HN-AD菌属能够在好氧和高C/N条件下快速脱氮,一方面是因为体系中充足的氧气及时补充了脱氮所需的电子受体;另一方面,高C/N保证了消耗碱度的及时补充,为脱氮提供了稳定的条件。HN-AD菌的这些生长特性使其在耐受高氨氮方面展现出了独特的优势[10]。表 4对可耐受和脱除高氨氮的HN-AD菌属进行了总结。如丛毛单胞菌Comamonas WXZ-17[69]可耐受初始浓度为817 mg/L的氨氮,氨氮和TN去除率分别达到了36.1%和26.3%;Shoda等[70]利用菌株产碱杆菌Alcaligenes No.4去除初始氨氮浓度约1 000 mg/L的硝化污泥,24 h内氨氮可彻底去除,平均氨氮去除速率达到了121 mg/(L·h),反硝化率达到了76.2%。对比可发现,不同种属的HN-AD菌株对氨氮的耐受程度及脱氮效果不同,如从活性污泥中筛选的镰刀菌Fusarium sp. A60[71]可耐受的初始氨氮浓度为800 mg/L,氨氮和TN去除率分别达到92%和88.4%,而从活性污泥中分离的不动杆菌Acinetobacter sp. Y1[72]耐受的初始氨氮浓度却能达到1 600 mg/L,同时对初始浓度为108.2 mg/L的氨氮去除率达到了98.8%,TN去除率达到了90.9%。此外,同属不同种的HN-AD菌的氨氮和TN脱除能力也存在差异,如不动杆菌Acinetobacter sp. TN-14[73]和不动杆菌Acinetobacter sp. SQ2[74]处理初始浓度约为600 mg/L的氨氮时,氨氮去除率分别为65.7%和97.8%,TN去除率分别为64.8%和73.2%。
| Highest tolerant ammonia nitrogen concentration (mg/L) | Genus | Strain | Ammonia removal or degradation | Total nitrogen removal (%) |
| 600.20 | Acinetobacter | TN-14 | 65.7% | 64.8 |
| 603.24 | Acinetobacter | SQ2 | 97.8% | 73.2 |
| 800.00 | Fusarium sp. | A60 | 92.0% | 88.4 |
| 871.00 | Comamonas | WXZ-17 | 36.1% | 26.3 |
| 897.29 | Bacillus cereus | WXZ-8 | 142.2 mg/(L·h)[76] | 44.5 |
| 1 121.24 | Bacillus methylotrophicus | L7 | 3.8 mg/(L·h) | Not mentioned |
| 1 200.00 | Alcaligenes | No.4 | 90.0% | Not mentioned |
| 1 600.00 | Acinetobacter sp. | Y1 | 98.0% | 90.9 |
| 2 000.00 | Zobellella taiwanensis | DN-7 | 19.6 mg/(L·h)[77] | 49.5 |
| 4 000.00 | Halomonas sp. | B01 | 99.2% | 96.0 |
通过对HN-AD菌属耐受高氨氮机制的解析,发现上述菌株具备高氨氮脱除的特性与其代谢通路和生存环境具有密切关系。一方面,HN-AD菌属可实现氨氮、NO3–-N、NO2–-N三氮的同步脱除,不仅缩短了脱氮周期,而且降低了NO2–-N积累对菌体产生的毒害作用[9];另一方面,HN-AD菌属属于异养需氧型微生物,氧气和基质的供应不仅加速了细胞的增殖分化,使其快速将氨氮同化为细胞成分,而且使细胞保持了较高的脱氮酶活性,从酶活层面加速了高氨氮的脱除[75]。
2.4 其他极端环境下HN-AD的脱氮研究HN-AD菌除了能耐受高盐、低温、高浓度氨氮等极端条件,还能耐受贫营养、过量重金属、强碱等其他极端条件。如Su等[78]筛选的不动杆菌Acinetobacter sp. SYF26在C/N为4.5时,氨氮去除率可达到93.6%;动胶菌属Zoogloea sp. N299在C/N为1.1的条件下,TN去除率可达到46.4%[79]。C/N决定了反硝化电子供体的丰富度,在低C/N比条件下,普通脱氮菌株因为体系电子供体不足,脱氮速率通常不同程度降低,而部分HN-AD菌株在电子供体不足的条件下仍能保持较好的脱氮活性,充分展现了其耐受贫营养的优势,未来将有望应用于地下水污水、水库水源污染等微污染水源的治理[80-81]。重金属元素是微生物生长活动代谢过程中必不可少的微量元素,微量金属离子对微生物的酶活具有一定的促进作用,但过量的重金属对微生物具有毒性[82]。研究发现一些HN-AD菌属对过量重金属也表现出了耐受能力,如王瑶等[83]发现粪产碱杆菌Alcaligenes faecalis C16能够耐受≥22.4 mg/L的Fe2+;He等[84]分离的铜绿假单胞菌Pseudomonas aeruginosa PCN-2在好氧条件下能同步还原硝酸盐和5 mg/L的Cr6+。利用微生物对重金属的吸附和积累作用,可以超量吸收转移一种或几种重金属,从而达到减少环境中重金属含量的目的,因此,后续有必要继续开展HN-AD菌株对重金属的吸附和积累研究。此外,少数HN-AD菌株还可在强碱环境下保持活性,如假单胞菌P. chengduensis ZPQ2[85]的最适pH为11,表现出明显的嗜碱性,传统自养硝化菌的最适pH为6–8,相较而言,HN-AD菌株具有更加宽泛的pH适应范围[86]。HN-AD菌属在这些极端条件仍能发挥其脱氮功能,这为极端条件下的废水处理提供了新渠道,也极大地拓宽了HN-AD技术的应用范围。
3 极端条件下HN-AD的工艺研究HN-AD菌属在极端条件下表现出的脱氮潜力使得越来越多的学者开始进行相关菌株的工艺研究。目前工艺研究的主要方式包括与反应器结合、菌株固定化、直接投加于处理系统中等,研究者主要通过简化工艺流程、控制工艺条件、优化工艺过程等来提高脱氮效率。当前极端条件的工艺研究实例主要集中在低温、高温、高盐、高氨氮等。如在低温条件下,Yao等[13]为了解决低温条件下污水处理厂脱氮效率低等问题,将耐寒的HN-AD菌群投加到反应器中进行生物强化,并通过逐渐增加溶解氧浓度,结果不仅缩短了硝化时间,而且成功提高了低温(10 ℃)条件下污水氨氮的去除效率;在高温条件下,杨云龙[87]等将曝气生物滤池与螯台球菌Chelatococcus daeguensis TAD1相结合,考察了菌株在高温下的HN-AD性能,脱氮过程中控制滤池温度为50 ℃,同时优化了溶解氧参数,当以硫酸铵和硝酸钠分别为唯一氮源时,菌株的氨氮和TN去除率在12 h内均达到了100%。又如在高盐条件下,苏兵[12]将嗜盐HN-AD菌株SF16用于牡蛎壳填料曝气生物滤池,控制盐度为3%、C/N为10、pH为7.5–9.5、氨氮浓度为28.1–35.1 mg/L时,氨氮去除率达到了97.1%,TN去除率达到了73.92%;在高氨氮条件下,Joo[88]利用粪产碱杆菌Alcaligenes faecalis No.4处理氨氮浓度为2 000 mg/L的猪场废水,最大氨氮脱除速率达到了30 mg/(L·h);课题组将前期分离的HN-AD菌株TAC-1以10%的接种量投加于单级三维结构生物转盘(3D-RBC),控制转速为34 r/min、水力停留时间(HRT)为24 h、进水化学需氧量(Chemical oxygen demand,COD)为5 000 mg/L、氨氮浓度为500 mg/L时,氨氮、总氮、COD的去除率分别达到了99.4%、79.5%、85.2%,研究结果也证实了HN-AD菌株进行工艺研究时具有同步脱除高氨氮和COD的效果。HN-AD菌株的工艺研究是未来开发HN-AD生物脱氮技术的必经之路,能为以后开发生物脱氮工艺提供重要的理论参数和实践经验。
4 总结与展望近20年来,关于HN-AD菌属的研究取得了许多实质性的进展,不仅加深了HN-AD菌属脱氮机理和脱氮特性的认识,而且挖掘了其在极端条件下的脱氮潜力。目前,约20多个属的HN-AD菌株被分离出来,一方面,研究者对其进行了生长特性、脱氮效果及代谢机理等方面的深入研究,发现HN-AD菌除了能耐受高盐、低温、高浓度氨氮等极端条件,还能耐受贫营养、过量重金属、强碱等其他极端条件。HN-AD菌属在这些极端条件仍能发挥其脱氮功能,为极端条件下废水的生物脱氮提供了新途径;另一方面,越来越多的学者开始了极端条件下菌株的工艺研究,主要方式包括结合反应器、菌株固定化、复配脱氮菌剂等,一定程度上拓宽了HN-AD菌的研究范围[8-10]。HN-AD菌属具有生长繁殖快、世代时间短、脱氮效率高等优点,这使其在极端条件废水处理方面具有广阔的应用前景,有望攻克传统处理工艺无法兼顾处理效率与经济适用两方面的瓶颈[15]。
未来,HN-AD菌的研究将主要围绕好氧脱氮技术如何服务工程应用而展开,可能的拓展领域包括以下几方面:1)系统开展基于培养基优化的菌剂发酵工艺研究,分离筛选在贫营养条件具有高脱氮活性的HN-AD菌株;2)充分利用多组学测序技术,全面解析HN-AD菌属的特征酶以及功能基因,进一步明晰HN-AD菌种同步脱氮的机理;3)深入研究HN-AD菌与其他脱氮微生物存在的竞争和融合关系,辨识不同种属间的互利共生作用,筛选适于工程应用的HN-AD功能菌剂;4)基于现有在单一极端条件下的研究成果,开展HN-AD菌同时在多种极端环境下的脱氮研究,并拓展其在生物除磷中的应用。
| [1] | Zhang L, Xu EG, Li YB, et al. Ecological risks posed by ammonia nitrogen (AN) and un-ionized ammonia (NH3) in seven major river systems of China. Chemosphere, 2018, 202: 136–144. DOI: 10.1016/j.chemosphere.2018.03.098 |
| [2] |
Cai J, Zheng P. Simultaneous removal of sulfide and nitrite by anaerobic bioprocess.
Chin J Biotech, 2009, 25(11): 1684–1689.
(in Chinese). 蔡靖, 郑平. 亚硝酸盐型同步厌氧生物脱氮除硫工艺的运行性能. 生物工程学报, 2009, 25(11): 1684-1689. DOI:10.3321/j.issn:1000-3061.2009.11.014 |
| [3] |
Zhang ZZ, Ji YX, Chen H, et al. Application and obstacles of ANAMMOX process.
Chin J Biotech, 2014, 30(12): 1804–1816.
(in Chinese). 张正哲, 姬玉欣, 陈辉, 等. 厌氧氨氧化工艺的应用现状和问题. 生物工程学报, 2014, 30(12): 1804-1816. |
| [4] | Vincent WF. Enigmatic microorganisms and life in extreme environments. J Palaeolimnol, 2002, 27(2): 285–286. DOI: 10.1023/A:1014237900498 |
| [5] | Glass C, Silverstein J. Denitrification of high-nitrate, high-salinity wastewater. Water Res, 1999, 33(1): 223–229. DOI: 10.1016/S0043-1354(98)00177-8 |
| [6] | Morling S, Plaza E. Biological nitrogen removal at low water temperatures-long term experience. Desalinat Water Treat, 2011, 25(1/3): 226–232. |
| [7] | Zhao B, Tian M, An Q, et al. Characteristics of a heterotrophic nitrogen removal bacterium and its potential application on treatment of ammonium-rich wastewater. Bioresour Technol, 2016, 226: 46–54. |
| [8] |
Yang T, Yang Y, Liu YX. Research progress and challenges of heterotrophic nitrification-aerobic denitrification.
Mocrobiol China, 2017, 44(9): 2213–2222.
(in Chinese). 杨婷, 杨娅, 刘玉香. 异养硝化-好氧反硝化的研究进展. 微生物学通报, 2017, 44(9): 2213-2222. |
| [9] | Castignetii D, Hollocher TC. Heterotrophic nitrification among denitrifiers. Appl Environ Microbiol, 1984, 47(4): 620–623. |
| [10] |
He H, Yu X, Han YT, et al. Research progress in the denitrification characteristics of heterotrophic nitrification-aerobic denitrification bacteria.
Ind Water Treat, 2017, 37(4): 12–17.
(in Chinese). 何环, 余萱, 韩亚涛, 等. 异养硝化好氧反硝化菌脱氮特性的研究进展. 工业水处理, 2017, 37(4): 12-17. |
| [11] | Lei Y, Wang YQ, Liu HJ, et al. A novel heterotrophic nitrifying and aerobic denitrifying bacterium, Zobellella taiwanensis DN-7, can remove high-strength ammonium. Appl Microbiol Biot, 2016, 100(9): 4219–4229. DOI: 10.1007/s00253-016-7290-5 |
| [12] | Duan JM, Fang HD, Su B, et al. Characterization of a halophilic heterotrophic nitrification-aerobic denitrification bacterium and its application on treatment of saline wastewater. Bioresour Technol, 2015, 179: 421–428. DOI: 10.1016/j.biortech.2014.12.057 |
| [13] | Yao S, Ni JR, Chen Q, et al. Enrichment and characterization of a bacteria consortium capable of heterotrophic nitrification and aerobic denitrification at low temperature. Bioresour Technol, 2013, 127: 151–157. DOI: 10.1016/j.biortech.2012.09.098 |
| [14] | Littleton HX, Daigger GT, Strom PF, et al. Simultaneous biological nutrient removal: evaluation of autotrophic denitrification, heterotrophic nitrification, and biological phosphorus removal in full-scale systems. Water Environ Res, 2003, 75(2): 138–150. DOI: 10.2175/106143003X140926 |
| [15] |
Weng ZH, Lv H, Zhou JT. Research progress in the denitrification performances of heterotrophic nitrifying-aerobic denitrifying bacteria.
Ind Water Treat, 2017, 37(3): 21–25.
(in Chinese). 翁梓航, 吕红, 周集体. 异养硝化-好氧反硝化菌的脱氮性能研究进展. 工业水处理, 2017, 37(3): 21-25. |
| [16] |
Su WY, Gao JF, Zhao HM. Research progress in heterotrophic nitrification-aerobic denitrification bacteria.
Ind Water Treat, 2013, 33(12): 1–5.
(in Chinese). 苏婉昀, 高俊发, 赵红梅. 异养硝化-好氧反硝化菌的研究进展. 工业水处理, 2013, 33(12): 1-5. |
| [17] |
He X, Lv J, He YJ, et al. Study progress on the mechanism of heterotrophic nitrification.
Acta Microbiol Sin, 2006, 46(5): 844–847.
(in Chinese). 何霞, 吕剑, 何义亮, 等. 异养硝化机理的研究进展. 微生物学报, 2006, 46(5): 844-847. DOI:10.3321/j.issn:0001-6209.2006.05.034 |
| [18] | Wehrfritz JM, Reilly A, Spiro S, et al. Purification of hydroxylamine oxidase from Thiosphaera pantotropha. FEBS Lett, 1993, 335(2): 246–250. DOI: 10.1016/0014-5793(93)80739-H |
| [19] | Ji B, Yang K, Zhu L, et al. Aerobic denitrification: A review of important advances of the last 30 years. Biotechnol Bioproc Eng, 2015, 20(4): 643–651. DOI: 10.1007/s12257-015-0009-0 |
| [20] |
Sun XM, Li QF, Zhang Y, et al. Phylogenetic analysis and nitrogen removal characteristics of a heterotrophic nitrifying-aerobic denitrifying bacteria strain from marine environment.
Acta Microbiol Sin, 2012, 52(6): 687–695.
(in Chinese). 孙雪梅, 李秋芬, 张艳, 等. 一株海水异养硝化-好氧反硝化菌系统发育及脱氮特性. 微生物学报, 2012, 52(6): 687-695. |
| [21] |
Si WG, Lv ZG, Xu C. Isolation of heterotrophic nitrifiers which can tolerate high concentration of ammonia-nitrogen and the optimization of their nitrogen removal efficiency in wastewater.
Environ Sci, 2011, 32(11): 3448–3454.
(in Chinese). 司文攻, 吕志刚, 许超. 耐受高浓度氨氮异养硝化菌的筛选及其脱氮条件优化. 环境科学, 2011, 32(11): 3448-3454. |
| [22] | Chen PZ, Li J, Li QX, et al. Simultaneous heterotrophic nitrification and aerobic denitrification by bacterium Rhodococcus sp. CPZ24. Bioresour Technol, 2012, 116: 266–270. DOI: 10.1016/j.biortech.2012.02.050 |
| [23] | Hen MX, Wang WC, Feng Y, et al. Impact resistance of different factors on ammonia removal by heterotrophic nitrification-aerobic denitrification bacterium Aeromonas sp. HN-02. Bioresour Technol, 2014, 167: 456–461. DOI: 10.1016/j.biortech.2014.06.001 |
| [24] | Zhao B, An Q, He YL, et al. N2O and N2 production during heterotrophic nitrification by Alcaligenes faecalis strain NR. Bioresour Technol, 2012, 116: 379–385. DOI: 10.1016/j.biortech.2012.03.113 |
| [25] |
Liu JF, Zhou T, Liu ZH, et al. Screening and identification of a new heterotrophic nitrifying bacteria and characterization of nitrification.
Environ Sci Technol, 2014, 37(8): 99–103.
(in Chinese). 刘杰凤, 周天, 刘正辉, 等. 一株新型异养硝化细菌的分离鉴定及硝化特性. 环境科学与技术, 2014, 37(8): 99-103. |
| [26] | Zhang JB, Wu PX, Hao B, et al. Heterotrophic nitrification and aerobic denitrification by the bacterium Pseudomonas stutzeri YZN-001. Bioresour Technol, 2011, 102(21): 9866–9869. DOI: 10.1016/j.biortech.2011.07.118 |
| [27] | Li CE, Yang JS, Wang X, et al. Removal of nitrogen by heterotrophic nitrification-aerobic denitrification of a phosphate accumulating bacterium Pseudomonas stutzeri YG-24. Bioresour Technol, 2015, 182: 18–25. DOI: 10.1016/j.biortech.2015.01.100 |
| [28] | Jin R, Liu T, Liu G, et al. Simultaneous heterotrophic nitrification and aerobic denitrification by the marine origin bacterium Pseudomonas sp. ADN-42. Appl Biochem Biotechnol, 2015, 175(4): 2000–2011. DOI: 10.1007/s12010-014-1406-0 |
| [29] | Huang XF, Li WG, Zhang DY, et al. Ammonium removal by a novel oligotrophic Acinetobacter sp. Y16 capable of heterotrophic nitrification-aerobic denitrification at low temperature. Bioresour Technol, 2013, 146: 44–50. DOI: 10.1016/j.biortech.2013.07.046 |
| [30] | Huang TL, He XX, Zhang HH, et al. Nitrogen removal characteristics of the heterotrophic nitrification-aerobic denitrification bacterium Acinetobacter sp. Sxf14. Appl Microbiol Biot, 2015, 21(2): 201–207. |
| [31] | Ren YX, Yang L, Liang X. The characteristics of a novel heterotrophic nitrifying and aerobic denitrifying bacterium, Acinetobacter junii YB. Bioresour Technol, 2014, 171: 1–9. DOI: 10.1016/j.biortech.2014.08.058 |
| [32] |
Xin YF, Qu XH, Yuan MD, et al. Isolation and identification of a heterotrophic nitrifying and aerobic denitrifying Acinetobacter sp. YF14 and its denitrification activity.
Acta Microbiol Sin, 2011, 51(12): 1646–1654.
(in Chinese). 辛玉峰, 曲晓华, 袁梦冬, 等. 一株异养硝化-反硝化不动杆菌的分离鉴定及脱氮活性. 微生物学报, 2011, 51(12): 1646-1654. |
| [33] |
Huang TL, Zhang LN, Zhang HH, et al. Screening and nitrogen removal characteristics of a heterotrophic nitrification-aerobic denitrification strain.
Ecol Environ Sci, 2015, 24(1): 113–120.
(in Chinese). 黄廷林, 张丽娜, 张海涵, 等. 一株贫营养异养硝化-好氧反硝化菌的筛选及脱氮特性. 生态环境学报, 2015, 24(1): 113-120. |
| [34] | Padhi SK, Tripathy S, Sen R, et al. Characterisation of heterotrophic nitrifying and aerobic denitrifying Klebsiella pneumoniae CF-S9 strain for bioremediation of wastewater. Int Biodeter Biodegr, 2013, 78: 67–73. DOI: 10.1016/j.ibiod.2013.01.001 |
| [35] | Chen Q, Ni JR. Ammonium removal by Agrobacterium sp. LAD9 capable of heterotrophic nitrification-aerobic denitrification. J Biosci Bioeng, 2012, 113(5): 619–623. DOI: 10.1016/j.jbiosc.2011.12.012 |
| [36] | Thite VS, Nerurkar AS. Physicochemical characterization of pectinase activity from Bacillus sp. and their accessory role in synergism with crude xylanase and commercial cellulase in enzyme cocktail mediated saccharification of agrowaste biomass. J Appl Microbiol, 2018, 124(5): 1147–1163. DOI: 10.1111/jam.2018.124.issue-5 |
| [37] | Sun ZY, Lv YK, Liu YX, et al. Removal of nitrogen by heterotrophic nitrification-aerobic denitrification of a novel metal resistant bacterium Cupriavidus sp. S1. Bioresour Technol, 2016, 220: 142–150. DOI: 10.1016/j.biortech.2016.07.110 |
| [38] | Lee KY, Kim KW, Park M, et al. Novel application of nanozeolite for radioactive cesium removal from high-salt wastewater. Water Res, 2016, 95: 134–141. DOI: 10.1016/j.watres.2016.02.052 |
| [39] | Eom H, Kim J, Kim S, et al. Treatment of saline wastewater containing a high concentration of salt using marine bacteria and aerobic granule sludge. J Environ Eng, 2018, 144(5): 1–8. |
| [40] | Ji L, Liu Y, Zhang YF, et al. Study on the treatment of high salinity oil waste water by reverse osmosis technology. Adv Mater Res, 2012, 418-420: 90–93. |
| [41] | Tokuz RY, Eckenfelder WW Jr. Wesley Eckenfelder Jrr. The effect of inorganic salts on the activated sludge process performance. Water Res, 1979, 13: 99–104. DOI: 10.1016/0043-1354(79)90260-4 |
| [42] |
Qu Y, Zhang PY, Yang RX, et al. Characteristics of removal nitrogen and halotolerancy of halotolerant heterotrophic nitrifying strain qy18 and moderately halophilic heterotrophic nitrifying strain gs2.
Mar Environ Sci, 2011, 30(3): 337–341.
(in Chinese). 曲洋, 张培玉, 杨瑞霞, 等. 耐盐异养硝化菌qy18和中度嗜盐异养硝化菌gs2的脱氮特性与耐盐性研究. 海洋环境科学, 2011, 30(3): 337-341. DOI:10.3969/j.issn.1007-6336.2011.03.008 |
| [43] | Woolard CR, Irvine RL. Biological treatment of hypersaline wastewater by a biofilm of halophilic bacteria. Water Environ Res, 1994, 66(3): 230–235. DOI: 10.2175/WER.66.3.8 |
| [44] |
Han YH, Zhang WX, Zhuang ZG, et al. Isolation and characterization of the salt-tolerant aerobic denitrifying bacterial strain A-13.
Acta Microbiol Sin, 2013, 53(1): 47–58.
(in Chinese). 韩永和, 章文贤, 庄志刚, 等. 耐盐好氧反硝化菌A-13菌株的分离鉴定及其反硝化特性. 微生物学报, 2013, 53(1): 47-58. |
| [45] | Zhang QL, Liu Y, Ai GM, et al. The characteristics of a novel heterotrophic nitrification-aerobic denitrification bacterium, Bacillus methylotrophicus strain L7. Bioresour Technol, 2012, 108: 35–44. DOI: 10.1016/j.biortech.2011.12.139 |
| [46] | Guo Y, Zhou XM, Li YG, et al. Heterotrophic nitrification and aerobic denitrification by a novel Halomonas campisalis. Biotechnol Lett, 2013, 35(12): 2045–2049. DOI: 10.1007/s10529-013-1294-3 |
| [47] | Tsuneda S, Mikami M, Kimochi Y, et al. Effect of salinity on nitrous oxide emission in the biological nitrogen removal process for industrial wastewater. J Hazard Mater, 2005, 119(1/3): 93–98. |
| [48] | Zheng HY, Liu Y, Gao XY, et al. Characterization of a marine origin aerobic nitrifying-denitrifying bacterium. J Biosci Bioeng, 2012, 114(1): 33–37. DOI: 10.1016/j.jbiosc.2012.02.025 |
| [49] | Yao RL, Qiu LN, Zhang WW, et al. Isolation and characteristics of heterotrophic nitrification-aerobic denitrification bacterium, Bacillus cereus X7 at high salinity. Adv Mater Res, 2014, 864-867: 111–114. |
| [50] | Wang T, Li J, Zhang LH, et al. Simultaneous heterotrophic nitrification and aerobic denitrification at high concentrations of NaCl and ammonia nitrogen by Halomonas bacteria. Water Sci Technol, 2017, 76(2): 386–395. DOI: 10.2166/wst.2017.214 |
| [51] | Halmø G, Eimhjellen K. Low temperature removal of nitrate by bacterial denitrification. Water Res, 1981, 15(8): 989–998. DOI: 10.1016/0043-1354(81)90207-4 |
| [52] |
Wei W, Huang TL. Isolation and identification of a low temperature and aerobic denitrifier and its denitrification characteristics.
Water Technol, 2012, 6(6): 12–16.
(in Chinese). 魏巍, 黄廷林. 低温好氧反硝化菌的分离鉴定及脱氮特性研究. 供水技术, 2012, 6(6): 12-16. DOI:10.3969/j.issn.1673-9353.2012.06.004 |
| [53] | Ye Q, Li KL, Li ZL, et al. Heterotrophic nitrification-aerobic denitrification performance of strain Y-12 under low temperature and high concentration of inorganic nitrogen conditions. Water, 2017, 9(11): 835–845. DOI: 10.3390/w9110835 |
| [54] | Qu D, Wang C, Wang YF, et al. Heterotrophic nitrification and aerobic denitrification by a novel groundwater origin cold-adapted bacterium at low temperatures. RSC Adv, 2014, 5(7): 5149–5157. |
| [55] | Zhang DY, Li WG, Huang XF, et al. Removal of ammonium in surface water at low temperature by a newly isolated Microbacterium sp. strain SFA13. Bioresour Technol, 2013, 137: 147–152. DOI: 10.1016/j.biortech.2013.03.094 |
| [56] | Dziewit L, Bartosik D. Plasmids of psychrophilic and psychrotolerant bacteria and their role in adaptation to cold environments. Front Microbiol, 2014, 5: 596. |
| [57] |
Feng Y. Research on the response mechanism of heterotrophic nitrification-aerobic denitrification bacterial HN-02 antioxidant enzyme system under low-temperature shock[D]. Chengdu: Southwest Jiaotong University, 2014 (in Chinese). 冯叶.低温冲击对异养硝化-好氧反硝化菌HN-02的影响研究[D].成都: 西南交通大学, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10613-1014257727.htm |
| [58] |
Ye BJ. Study of the microbial metabolic activities by the Microcalorimetric method[D]. Lanzhou: Lanzhou University, 2008 (in Chinese). 叶丙静.微生物代谢活性的微量热研究[D].兰州: 兰州大学, 2008. http://cdmd.cnki.com.cn/Article/CDMD-10730-2008162744.htm |
| [59] |
Liu B. Research and application on thermal and mass transfer in the integrated using of biologic substance[D]. Chongqing: Chongqing University, 2008 (in Chinese). 刘彬.有机物综合利用中的传热传质分析与应用[D].重庆: 重庆大学, 2008. http://cdmd.cnki.com.cn/Article/CDMD-10611-2009050700.htm |
| [60] |
Liu J. Biological character research of the activated sludge/biofilm process treating Municiple wastewater in North area[D]. Harbin: Harbin Institute of Technology, 2006 (in Chinese). 刘杰.活性污泥/生物膜工艺处理北方城市污水微生物特性研究[D].哈尔滨: 哈尔滨工业大学, 2006. http://cdmd.cnki.com.cn/Article/CDMD-10213-2007033667.htm |
| [61] |
Jiang W, Li J, Meng LQ, et al. Effect of environmental factors on ammonium removal of Pseudomonas fluorescens wsw-1001 at low temperature.
Environ Sci Technol, 2014, 37(1): 27–30.
(in Chinese). 姜威, 李晶, 孟利强, 等. 环境因素对荧光假单胞杆菌低温脱氮的影响. 环境科学与技术, 2014, 37(1): 27-30. |
| [62] |
He TX, Ni JP, Li ZL, et al. Heterotrophic nitrification and aerobic denitrification of the hypothermia aerobic denitrification bacterium: Arthrobacter arilaitensis.
Environ Sci, 2016, 37(3): 1082–1088.
(in Chinese). 何腾霞, 倪九派, 李振轮, 等. 1株Arthrobacter arilaitensis菌的耐冷异养硝化和好氧反硝化作用. 环境科学, 2016, 37(3): 1082-1088. |
| [63] | Yi X, He TX, Li ZL, et al. Nitrogen removal characteristics of Pseudomonas putida Y-9 capable of heterotrophic nitrification and aerobic denitrification at low temperature. Biomed Res Int, 2017, 2017: 1429018. |
| [64] |
He Y, Zhao YC, Zhou GM. Research progress on the denitrogenation of highly concentrated ammonium-nitrogen wastewater.
Ind Water Treat, 2008, 28(1): 1–4.
(in Chinese). 何岩, 赵由才, 周恭明. 高浓度氨氮废水脱氮技术研究进展. 工业水处理, 2008, 28(1): 1-4. DOI:10.3969/j.issn.1005-829X.2008.01.001 |
| [65] |
Liu YM, Hao ZL. Treatment technologies and research status of highly concentrated ammonium-nitrogen wastewater.
Technol Water Treat, 2012, 38(S1): 7–11.
(in Chinese). 刘亚敏, 郝卓莉. 高氨氮废水处理技术及研究现状. 水处理技术, 2012, 38(S1): 7-11. |
| [66] | Vadivelu VM, Keller J, Yuan Z. Effect of free ammonia and free nitrous acid concentration on the anabolic and catabolic processes of an enriched nitrosomonas culture. Biotechnol Bioeng, 2010, 95(5): 830–839. |
| [67] | Ciudad G, Rubilar O, Muñoz P, et al. Partial nitrification of high ammonia concentration wastewater as a part of a shortcut biological nitrogen removal process. Process Biochem, 2005, 40(5): 1715–1719. DOI: 10.1016/j.procbio.2004.06.058 |
| [68] |
Wang JF, Tu BH, Chen RP, et al. Application and development of new biological nutrient removal technology.
Techniq Equip Environ Pollut ControlTechniq Equip Environ Pollut Control, 2003, 4(9): 70–73.
(in Chinese). 王建芳, 涂宝华, 陈荣平, 等. 生物脱氮除磷新工艺的研究进展. 环境污染治理技术与设备, 2003, 4(9): 70-73. |
| [69] |
Liao XH. Study on the optimizing ability and gas production of heterotrophic nitrification-aerobic denitrification strains[D]. Beijing: Beijing Technology and Business University, 2009 (in Chinese). 廖小红.异养硝化-好氧反硝化菌优选及脱氮产物研究[D].北京: 北京工商大学, 2009. http://cdmd.cnki.com.cn/Article/CDMD-10011-2010044305.htm |
| [70] | Shoda M, Ishikawa Y. Heterotrophic nitrification and aerobic denitrification of high-strength ammonium in anaerobically digested sludge by Alcaligenes faecalis strain No. 4. J Biosci Bioeng, 2014, 117(6): 737–741. DOI: 10.1016/j.jbiosc.2013.11.018 |
| [71] |
Yang XH. Isolation and characteristics study of a heterotrophic nitrification-aerobic denitrification fungus[D]. Shanxi: Taiyuan University of Technology, 2012 (in Chinese). 杨晓华.一株异养硝化好氧反硝化真菌的筛选及特性研究[D].山西: 太原理工大学, 2012. http://cdmd.cnki.com.cn/Article/CDMD-10112-1016025055.htm |
| [72] |
Song YJ, Li Y, Liu YX, et al. Effect of carbon and nitrogen sources on nitrogen removal by a heterotrophic nitrification-aerobic denitrification strain Y1.
Acta Sci Circumst, 2013, 33(9): 2491–2497.
(in Chinese). 宋宇杰, 李屹, 刘玉香, 等. 碳源和氮源对异养硝化好氧反硝化菌株Y1脱氮性能的影响. 环境科学学报, 2013, 33(9): 2491-2497. |
| [73] |
Xin X, Yao L, Lu L, et al. Identification of a high ammonia nitrogen tolerant and heterotrophic nitrification-aerobic denitrification bacterial strain TN-14 and its nitrogen removal capabilities.
Environ Sci, 2014, 35(10): 3926–3932.
(in Chinese). 信欣, 姚力, 鲁磊, 等. 耐高氨氮异养硝化-好氧反硝化菌TN-14的鉴定及其脱氮性能. 环境科学, 2014, 35(10): 3926-3932. |
| [74] |
Wang TY, Wei HF, Hu ZQ, et al. Isolation and identification of a heterotrophic nitrifying and aerobic denitrifying strain and its denitrification characteristics.
Acta Sci Circumst, 2017, 37(3): 945–953.
(in Chinese). 王田野, 魏荷芬, 胡子全, 等. 一株异养硝化好氧反硝化菌的筛选鉴定及其脱氮特性. 环境科学学报, 2017, 37(3): 945-953. |
| [75] |
Yu DY, Zhang LY, Gao B. Factors affecting the heterotrophic nitrification property of heterotrophic nitrification-aerobic denitrifier.
Chem Ind Eng Prog, 2012, 31(12): 2797–2800.
(in Chinese). 于大禹, 张琳颖, 高波. 异养硝化-好氧反硝化菌异养硝化性能的影响因素. 化工进展, 2012, 31(12): 2797-2800. |
| [76] |
Liao XH, Wang P, Diao HF, et al. Heterotrophic nitrification-aerobic denitrification ability of Bacillus cereus WXZ-8.
Environ Poll Control, 2009, 31(7): 17–20.
(in Chinese). 廖小红, 汪苹, 刁惠芳, 等. 蜡状芽孢杆菌WXZ-8的异养硝化/好氧反硝化性能研究. 环境污染与防治, 2009, 31(7): 17-20. DOI:10.3969/j.issn.1001-3865.2009.07.006 |
| [77] |
Xiao JB, Jiang HX, Chu SY. Isolation and identification of aerobic denitrifying bacterium Defluvibacter lusatiensis strain DN7 and its heterotrophic nitrification ability.
Chin J Appl Ecol, 2012, 23(7): 1979–1984.
(in Chinese). 肖继波, 江惠霞, 褚淑祎. 好氧反硝化菌Defluvibacter lusatiensis str. DN7的分离鉴定和异养硝化性能. 应用生态学报, 2012, 23(7): 1979-1984. |
| [78] | Su JF, Zhang K, Huang TL, et al. Heterotrophic nitrification and aerobic denitrification at low nutrient conditions by a newly isolated bacterium, Acinetobacter sp. SYF26. Microbiology,, 2015, 161(4): 839–837. |
| [79] | Huang TL, Zhou SL, Zhang HH, et al. Nitrogen removal characteristics of a newly isolated indigenous aerobic denitrifier from oligotrophic drinking water reservoir, Zoogloea sp. N299. Int J Mol Sci, 2015, 16(5): 10038–10060. |
| [80] |
Li JD. Study on carbon source supplement for denitrification[D]. Shanghai: Tongji University, 2007 (in Chinese). 李基东.反硝化脱氮补充碳源选择与研究[D].上海: 同济大学, 2007. http://cdmd.cnki.com.cn/Article/CDMD-10247-2008050748.htm |
| [81] | Wang HY, Song Q, Wang J, et al. Simultaneous nitrification, denitrification and phosphorus removal in an aerobic granular sludge sequencing batch reactor with high dissolved oxygen: Effects of carbon to nitrogen ratios. Sci Total Environ, 2018, 642: 1145–1152. DOI: 10.1016/j.scitotenv.2018.06.081 |
| [82] | Giller KE, Witter E, Mcgrath SP. Heavy metals and soil microbes. Soil Biol Biochem, 2009, 41(10): 2031–2037. DOI: 10.1016/j.soilbio.2009.04.026 |
| [83] |
Wang Y, Liu YX, An H, et al. Influence of metal ions on nitrogen removal and NO2--N accumulation by Alcaligenes faecalis C16.
Mocrobiology, 2014, 41(11): 2254–2263.
(in Chinese). 王瑶, 刘玉香, 安华, 等. 金属离子对粪产碱杆菌C16的脱氮和亚硝酸盐积累的影响. 微生物学通报, 2014, 41(11): 2254-2263. |
| [84] | He D, Zheng MS, Ma T, et al. Interaction of Cr(Ⅵ) reduction and denitrification by strain Pseudomonas aeruginosa PCN-2 under aerobic conditions. Bioresour Technol, 2015, 185: 346–352. DOI: 10.1016/j.biortech.2015.02.109 |
| [85] | Kim JK, Park KJ, Cho KS, et al. Aerobic nitrification-denitrification by heterotrophic Bacillus strains. Bioresour Technol, 2005, 96(17): 1897–906. DOI: 10.1016/j.biortech.2005.01.040 |
| [86] |
Pan YJ, Liu F, Meng S, et al. Isolation and characterization of an aerobic denitrifying-heterotrophic bacterium.
Chin J Environ Eng, 2016, 34(1): 41–46.
(in Chinese). 潘玉瑾, 刘芳, 孟爽, 等. 好氧反硝化菌P. chengduensis ZPQ2的筛选及其反硝化条件优化. 环境工程, 2016, 34(1): 41-46. |
| [87] | Yang YL, Huang SB, Zhang YQ, et al. Nitrogen removal by Chelatococcus daeguensis TAD1 and its denitrification gene identification. Appl Biochem Biotechnol, 2014, 172(2): 829–839. DOI: 10.1007/s12010-013-0590-7 |
| [88] | Joo HS, Hirai M, Shoda M. Piggery wastewater treatment using Alcaligenes faecalis strain No.4 with heterotrophic nitrification and aerobic denitrification. Water Res, 2006, 40(16): 3029–3036. DOI: 10.1016/j.watres.2006.06.021 |
 2019, Vol. 35
2019, Vol. 35